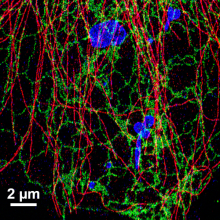
Single-molecule localization microscopy methods for super-resolution fluorescence microscopy such as STORM (stochastic optical reconstruction microscopy) are generally limited to thin three-dimensional (3D) sections (<600 nm) because of photobleaching of molecules outside the focal plane. Although multiple focal planes may be imaged before photobleaching by focusing progressively deeper within the sample, image quality is compromised in this approach because the total number of measurable localizations is divided between detection planes. Here, we solve this problem on fixed samples by developing an imaging method that we call probe-refresh STORM (prSTORM), which allows bleached fluorophores to be straightforwardly replaced with nonbleached fluorophores. We accomplish this by immunostaining the sample with DNA-conjugated antibodies and then reading out their distribution using fluorescently-labeled DNA-reporter oligonucleotides that can be fully replaced in successive rounds of imaging. We demonstrate that prSTORM can acquire 3D images over extended depths without sacrificing the density of localizations at any given plane. We also show that prSTORM can be adapted to obtain high-quality, 3D multichannel images with extended depth that would be challenging or impossible to achieve using established probe methods.